kit test Covid-19 IgG/IgM Rapid
Supplier : Monoïde Bec Consulting (Algeria)
Medical Equipment
Para-pharmacy and pharmacy
Biotechnologies
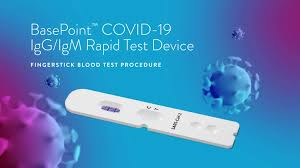
For rapid detection of IgG and IgM anti?SARS?CoV?2 (2019?nCoV) in 10?15 minutes during Covid?19 infection COVID?19 (Corona Virus Disease) is the most recently discovered infectious disease caused by the coronavirus, SARS?CoV?2 (2019?nCoV). The rapid test for the diagnosis of SARS?CoV?2 allows qualitative detection of IgG and/or IgM in human serum, total blood or plasma in about 10 to 15 minutes. Rapid tests are based on the principle of lateral flux immunochromatography and are available as a cassette. The test is based on separating the components of a mixture through a medium using the hair force and the specific and rapid binding of an antibody to its antigen. IgM and IgG are immunoglobulins produced by the immune system to protect against SARS?CoV?2. IgM and IgG anti?SARS?CoV?2 can therefore be detected in samples of affected patients. What are the benefits of quick testing? ? Quick results in 10?15 minutes. ? High detection efficiency: simultaneous monitoring of IgM and IgG. ? Detection without any test equipment. ? Easy to use, it is compatible with serum/total blood/plasma. ? Storage at room temperature. What is the principle of rapid testing for SARS?CoV?2 The test detects the presence of patient?generated antibodies against SARS?CoV?2, the virus that causes COVID?19 disease. The test can detect two types of antibody isotypes: IgG and IgM. There are several types of tests but the most common is to attach human anti?IgG and IgM antibodies to the surface of the cassette and pair a virus antigen with colloidal gold particles. If the patient's sample contains anti?SARS?CoV?2 antibodies then these antibodies will attach to the antigen present in the conjugation zone of the cassette and the formed complex will migrate to the human anti?IgG and/or IgM attached to the membrane. A coloured strip will then appear (see the various possible results below). The level of IgM antibodies begins to increase about a week after the initial infection, while IgG appears later than IgM (usually within 14 days of infection) and can last 6 months or even several years, meaning that IgGs serve as an indicator of a previous infection. Patients who are infected with SARSCoV? 2 can be quickly identified by simultaneous monitoring of IgM and IgG. During the 2003 SARS and Zika 2016 outbreak period, IgM/IgG antibody detection was used as one of the recommended diagnostic methods. A sample may be positive if IgM and/or IgG antibodies are present. There are different tapes for quick tests. In general, for the qualitative detection of IgG and IgM at the same time, there are three different lines: one for IgG, one for IgM and one for control. To be validated, this test must have a positive line for control (C) Results Interpretation IgM+ / IgG+ recent SARS?CoV?2 infection IgM+ / IgGrecent SARS?CoV?2 infection IgM? / IgG+ Pre?SARS?CoV?2 Infection IgM? / IgGNo infection or detectable antibodies during the onset of infection Medical devices for in vitrodiagnosis. Read the instructions carefully. All of these kits are intended for in vitro diagnosis and can only be used by healthcare professionals.
Other product information
Contacter par WhatsApp
Contact Supplier'